New Insights into Getting Older: Emerging Perspectives on Aging in Pets and People

The world’s human population is aging at an unprecedented pace. Our dogs and cats are also growing older, right by our sides. Although medical advances have helped increase lifespans for both people and pets, aging continues to be the biggest risk factor for age-related diseases. We are all getting older, but not necessarily healthier.

We have to fight against aging, as we do against a disease.
Cicero's De Senectute (Cato Maior): On Old Age
Age itself has often been considered a disease, something to be treated separately from age-related diseases such as cognitive decline. However, that viewpoint is changing. Research shows that while age is a major driver for many diseases, disease can also accelerate aging. Although solutions to age-related diseases are important, finding ways to delay aging could avoid a whole range of age-related illness. Slowing the aging process could extend the spans of life and health.1–4
One consistently proven nutritional approach that can modulate the effects of aging is caloric restriction. Defined as a reduction in nutrient availability – without malnutrition – hundreds of studies have shown that a regimen of reduced calories can increase lifespan and delay the onset of multiple age-related morbidities.2,5–8 In dogs, for example, a 25% reduction in calories over the course of a lifetime led to about 2 years’ increase in median lifespan for the controlled-fed dogs.9 Although studies on dietary restriction have been conducted for more than half a century, scientists are still actively researching the molecular mechanisms that explain these results.
From the Outside

In the lifetime study of dogs on restricted calorie diets, as all the dogs aged, those in the restricted-feeding group had a significantly greater mean percentage of lean body mass (LBM).9 Research in humans, dogs, and cats, has repeatedly shown that maintaining LBM is important: loss of LBM is associated with greater risk of mortality.9–15
Wasting of muscle mass is part of the LBM loss that comes with aging. Humans lose muscle at an estimated rate of 1-2% per year after 50 years of age.16 Studies in humans show that muscle fibers change in shape, structure and metabolism with age. The decline of muscle mass, called sarcopenia, has significant health impacts beyond diminished strength and function. Sarcopenia is a key component of “frailty”, considered a negative prognostic indicator for all-cause of mortality in elderly people.12,13,17–19
Although frailty is not a common term in veterinary practice, the impact is easy to see when senior dogs struggle to rise from lying down positions or climb stairs. While veterinarians can track this common aging condition by including muscle condition scores with every physical exam, long-term studies in people may be able to identify the factors that influence sarcopenia and improve understanding of muscle loss in dogs and cats.

Research has shown that nutritional intervention may help stave off muscle loss in elderly people as well as dogs and cats. It takes protein to build and maintain muscle. Yet, with age, people and pets often do not get enough dietary protein. In addition, studies show that protein synthesis and protein digestibility may decline with advancing years.20–22 Although age-associated muscle losses cannot be avoided entirely, increased dietary protein may stimulate protein synthesis and help preserve lean body mass.23,24
Precisely how much protein is appropriate for elderly humans and pets is a controversial subject. In companion animals, there are no Association of American Feed Control Officials (AAFCO) standards for senior pets. The AAFCO assessments for protein requirements are based on the amount of protein needed to maintain a balance between nitrogen intake (dietary protein) and nitrogen loss in stool and urine. However, multiple studies indicate that the amount of protein required to maintain LBM is much higher than that needed to maintain nitrogen balance.24,25
More recently, scientists found that polyunsaturated fatty acids (PUFAs) from fish oils might also have a role in preventing sarcopenia. Studies of healthy, older women enrolled in an exercise training program showed those who ingested 2-4 grams of fish oil daily for 3 months had greater gains in muscle strength compared with women who did not take fish oil.26,27
Changes in energy needs for maintenance and fat digestibility also affect age-related weight loss. Both dogs and cats may experience a decline in fat digestibility with age. However, while senior dogs require less energy for maintenance needs, cats have an increased demand for energy after about 13 years of age.28
All too often, comorbidities such as chronic kidney disease, enteropathy, or cancer, complicate weight loss in senior pets. Antioxidants, vitamins, and acidosis have all been shown to have roles in nutritionally managing these age-related diseases. Even when pets have multiple health concerns that could create conflicting management recommendations, routine nutritional assessments can help track pets’ changing dietary needs.
Aging on the Inside
Preventive health is a key component of senior care programs for both humans and pets. The challenge is early disease detection, so that intervention can begin before detrimental changes occur.
With advances in technology – particularly the “omics” that enable study of an individual’s DNA and its translation into cellular action – researchers can probe the molecular level impact of changes in nutrition.
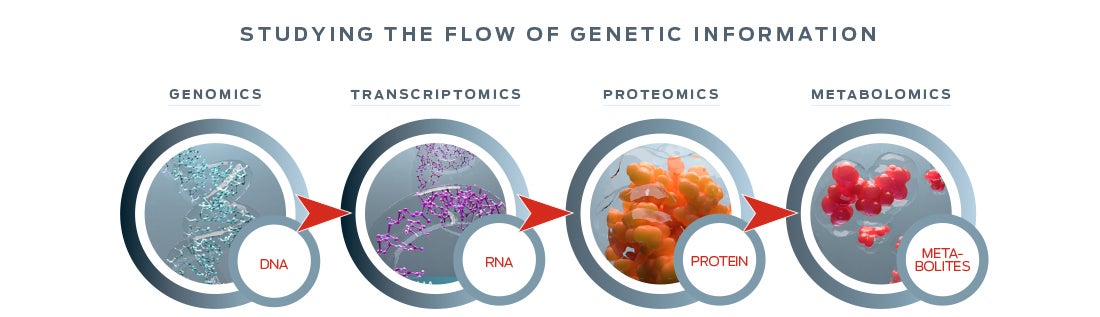
Omics research helped reveal how dietary restriction modulates cell activity in ways that are linked with longevity and healthspan.29 One factor that appears to play a central role in aging is the target of rapamycin (TOR) pathway, which regulates protein synthesis in every cell. When nutrients are depleted, mTOR activity is reduced. This results in a cascade of cellular responses that promote longevity and enhance resistance to stress.30,31
Scientists also use omics to search for reliable biomarkers of aging. Ideally, such biomarkers could predict health outcomes and track how nutritional interventions delay the onset of age- related diseases. For example, an individual’s metabotype – the profile of an individual’s microbiome and metabolome – could provide information that ultimately leads to a personalized nutritional prescription.32
Another potential biomarker for aging may be a mitochondrial protein called translocator protein (TSPO). Mitochondria not only produce energy for cells, they can also influence cell death. Faulty mitochondrial regulation is linked with many age-related diseases, such as Alzheimer’s disease. TSPO is important in mitochondrial regulation, and its function is dependent upon dietary cholesterol levels.33,34 In the future, developing methods to track TSPO, in clinics, could provide a way to measure health at the cellular level, assess the effect of diet, and lead to predictors for diseases related to mitochondrial dysfunction.
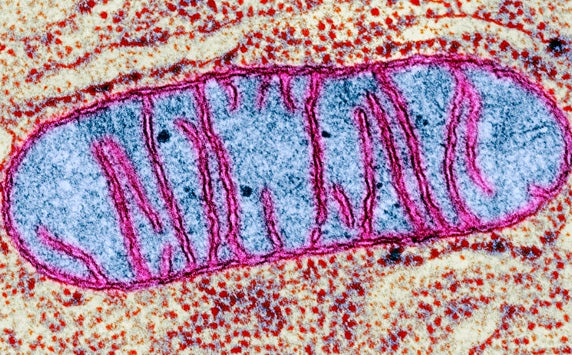
Omics also offer a way to look at molecular-level differences between the young and the old. Such differences can point scientists toward metabolic pathways that drive aging changes – and toward nutritional interventions that may improve health in senior pets.35
With omics, even microbial populations in the gastrointestinal (GI) tract can yield clues on healthy aging. More than a trillion GI bacteria form networks that sends signals throughout the body. Early studies of bacterial populations showed that both older people and pets have decreased fecal concentrations of beneficial bacteria such as bifidobacteria and increased concentrations of potentially harmful bacteria.36–38
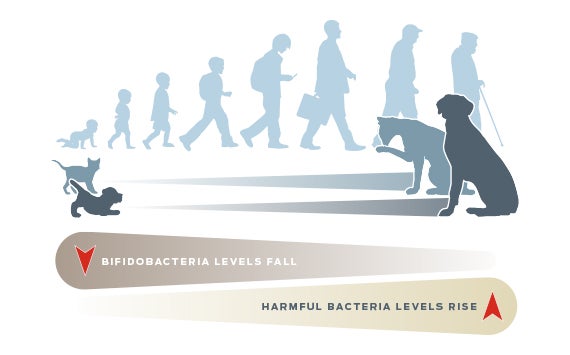
More recently, metagenomic studies, which profile entire populations of microbial DNA, have linked the intestinal microbiome with ever-expanding effects on brain health, longevity, skin conditions – even the effectiveness of cancer treatments.39,40
Such studies make it seem as if anti-aging solutions are almost within reach. While advances in medicine and technology have created new opportunities for understanding aging at the molecular level, there is no baseline understanding of what healthy aging looks like.
In an effort to fill that information gap, researchers at the University of Washington and Texas A&M University are collaborating on a Dog Aging Project (DAP) based in the U.S. This longitudinal study is planned to enroll 10,000 companion dogs to develop measures of aging and create a model of healthy aging with the power to predict, diagnose, treat and prevent age-related disease. With these defined measurements, practitioners will have new tools to help make their patients’ later years of life as healthy as possible.41,42
With a citizen-science approach, the DAP is designed to draw in not only veterinarians, but also dog-loving clients. The study offers opportunities for veterinarians to help develop new practice-based tools, and have more conversations with clients about age-related issues.
As geroscience advances, so will the possibilities for nutrition to positively influence the lives of our pets and, perhaps, ourselves.
References
-
Franceschi, C., Garagnani, P., Morsiani, C., Conte, M., Santoro, A., Grignolio, A., Salvioli, S. (2018). The continuum of aging and age-related diseases: Common mechanisms but different rates. Frontiers in Medicine, 5, 61. doi: 10.3389/fmed.2018.00061
-
Kaeberlein, M., Rabinovitch, P. S., & Martin, G. M. (2015). Healthy aging: The ultimate preventative medicine. Science, 350, 1191–1193. doi: 10.1126/science.aad3267
-
Kennedy, B. K., Berger, S. L., Brunet, A., Campisi, J., Cuervo, A. M., Epel, E. S., Sierra, F. (2014). Aging: a common driver of chronic diseases and a target for novel interventions. Cell, 159(4), 709–713. doi:10.1016/j.cell.2014.10.039
-
Olshansky, S. J., Perry, D., Miller, R. A., & Butler, R. N. (2006). In pursuit of the longevity dividend. The Scientist, 20, 28–36.
-
Kennedy, B. K., Steffen, K. K., & Kaeberlein, M. (2007). Ruminations on dietary restriction and aging. Cellular and Molecular Life Sciences, 64, 1323—1328. doi: 10.1007/s00018-007-6470-y
-
Kirkland, J. L. (2013). Translating advances from the basic biology of aging into clinical application. Experimental Gerontology, 48(1), 1–5. doi: 10.1016/j.exger.2012.11.014
-
Most, J., Tosti, V., Redman, L. M., & Fontana, L. (2017). Calorie restriction in humans: An update. Ageing Research Review, 39, 36—45. doi: 10.1016/j.arr.2016.08.005
-
Omodei, D., & Fontana, L. (2011). Calorie restriction and prevention of age-associated chronic disease. Federation of European Biochemical Societies Letters, 585(11), 1537–1542.
-
Kealy, R. D., Lawler, D. F., Ballam, J. M., Mantz, S. L., Biery, D. N., Greeley, E. H., Lust, G., Segre, M., Stowe, H.D. (2002). Effects of diet restriction on life span and age-related changes in dogs. Journal of the American Veterinary Medical Association, 220(9), 1315–1320. doi:10.2460/javma.2002.220.1315
-
Adams, V. J., Watson, P., Carmichael, S., Gerry, S., Penell, J., & Morgan, D. M. (2016). Exceptional longevity and potential determinants of successful ageing in a cohort of 39 Labrador retrievers: results of a prospective longitudinal study. Acta Veterinaria Scandinavica, 58, 29. doi: 10.1186/s13028-016-0206-7
-
Freeman, L. M. (2012). Cachexia and sarcopenia: Emerging syndromes of importance in dogs and cats. Journal of Veterinary Internal Medicine, 26, 3—17.
-
Han, S. S., Kim, K. W., Kim, K., Na, K. Y., Chae, D., Kim, S. & Chin, H.J. (2010). Lean Mass index: A better predictor of mortality than body mass index in elderly Asians. Journal of the American Geriatrics Society, 58 (2), 312—317. doi: 10.1111/j.1532-5415.2009.02672.x
-
Szulc, P., Munoz, F., Marchand, F., Chapurlat, R., & Delmas, P. D. (2010). Rapid loss of appendicular skeletal muscle mass is associated with higher all-cause mortality in older men: the prospective MINOS study. The American Journal of Clinical Nutrition, 91 (5), 1227–1236. doi:10.3945/ajcn.2009.28256
-
Teng, K. T., McGreevy, P. D., Toribio, J. L., Raubenheimer, D., Kendall, K., & Dhand, N. K. (2018). Strong associations of 9-point body condition scoring with survival and lifespan in cats. Journal of Feline Medicine and Surgery, 1:1098612X17752198. doi: 10.1177/1098612X17752198
-
Toss, F., Wiklund, P., Nordström, P., & Nordström, A. (2012). Body composition and mortality risk in later life. Age and Ageing, 41(5), 677–681. doi:10.1093/ageing/afs087
-
Buford, T. W., Anton, S. D., Judge, A. R., Marzetti, E., Wohlgemuth, S. E., Carter, C. S., Manini, T. M. (2010). Models of accelerated sarcopenia: Critical pieces for solving the puzzle of age-related muscle atrophy. Ageing Research Reviews, 9(4), 369–383. doi: 10.1016/j.arr.2010.04.004
-
Hua, J., Hoummady, S., Muller, C., Pouchelon, J., Blondot, M., Gilbert, C., & Desquilbet, L. (2016). Assessment of frailty in aged dogs. American Journal of Veterinary Research, 77(12), 1357—1365. doi: 10.2460/ajvr.77.12.1357
-
Song, M., Hu, F. B., Wu, K., Must, A., Chan, A. T., Willett, W. C., & Giovannucci, E. L. (2016). Trajectory of body shape in early and middle life and all cause and cause specific mortality: results from two prospective US cohort studies. The British Medical Journal, 353, 2195. doi:10.1136/bmj.i2195
-
Wolfe, R. R. (2006). The underappreciated role of muscle in health and disease. American Journal of Clinical Nutrition, 84(3), 475—482.
-
Laflamme, D. P., & Hannah, S. S. (2013). Discrepancy between use of lean body mass or nitrogen balance to determine protein requirements for adult cats. Journal of Feline Medicine and Surgery, 15(8), 691—697. doi: 10.1177/1098612X12474448
-
Robinson, S. M., Reginster, J. Y., Rizzoli, R., Shaw, S. C., Kanis, J. A., Bautmans, I., Bischoff-Ferrari, H., Cooper, C. (2018). Does nutrition play a role in the prevention and management of sarcopenia? Clinical Nutrition, 37(4), 1121—1132.
-
Wall, B. T., Gorissen, S. H., Pennings, B., Koopman, R., Groen, B. B. L., Verdijk, L. B., & van Loon, L. J. C. (2015). Aging Is accompanied by a blunted muscle protein synthetic response to protein ingestion. PloS ONE, 10(11), e0140903. Doi:10.1371/journal.pone.0140903
-
Deutz, N. E. P., Bauer, J. M., Barazzoni, R., Biolo, G., Boirie, Y., Bosy-Westphal, A., Calder, P. C. (2014). Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clinical Nutrition (Edinburgh, Scotland), 33(6), 929–936. doi: 10.1016/j.clnu.2014.04.007
-
Laflamme, D. P., & Hannah, S. S. (2005). Increased Dietary Protein promotes fat loss and reduces loss of lean body mass during weight loss in cats. International Journal of Applied Research in Veterinary Medicine, 3(2), 62—68
-
Wolfe, R. R., Miller, S.L., & Miller, K. B. (2008). Optimal protein intake in the elderly. Clinical Nutrition, 27(5), 675e84. doi: 10.1016/j.clnu.2008.06.008
-
Da Boit, M., Sibson, R., Sivasubramaniam, S., Meakin, J. R., Greig, C. A., Aspden, R. M., Gray, S. R. (2017). Sex differences in the effect of fish-oil supplementation on the adaptive response to resistance exercise training in older people: a randomized controlled trial. The American Journal of Clinical Nutrition, 105(1), 151–158. doi: 10.3945/ajcn.116.140780
-
Rodacki, C. L. N., Rodacki, A. L. F., Pereira, G., Naliwaiko, K., Coelho, I., Pequito, D., & Fernandes, L. C. (2012). Fish-oil supplementation enhances the effects of strength training in elderly women, The American Journal of Clinical Nutrition, 95(2), 428–436. doi: 10.3945/ajcn.111.021915
-
Laflamme, D. P., & Gunn-Moore, D. (2014). Nutrition of aging cats. Veterinary Clinics of North America: Small Animal Practice, 44, (4), 761—774. doi:10.1016/j.cvsm.2014.03.001
-
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2013). The hallmarks of aging. Cell, 153(6), 1194–1217. doi: 10.1016/j.cell.2013.05.039
-
Kapahi, P., Chen, D., Rogers, A. N., Katewa, S. D., Li, P. W. L., Thomas, E. L., & Kockel, L. (2010). With TOR less is more: a key role for the conserved nutrient sensing TOR pathway in aging. Cell Metabolism, 11(6), 453–465. doi: 10.1016/j.cmet.2010.05.001
-
Stanfel, M. N., Shamieh, L. S., Kaeberlein, M., & Kennedy, B. K. (2009). The TOR pathway comes of age. Biochimica et Biophysica Acta, 1790(10), 1067–1074. doi: 10.1016/j.bbagen.2009.06.007
-
Richards, S. E., Wang, Y., Claus, S. P., Lawler, D., Kochhar, S., Holmes, E., & Nicholson, J. K. (2013). Metabolic Phenotype Modulation by Caloric Restriction in a Lifelong Dog Study. Journal of Proteome Research, 12 (7), 3117—3127. doi:10.1021/pr301097k
-
Gatliff, J., & Campanella, M. (2012). The 18 kDa translocator protein (TSPO): a new perspective in mitochondrial biology. Current Molecular Medicine, 12(4), 356—368.
-
Gatliff, J., & Campanella, M. (2016). TSPO: kaleidoscopic 18-kDa amid biochemical pharmacology, control and targeting of mitochondria. Biochemical Journal, 473(2), 107—121. doi: 10.1042/BJ20150899
-
Swanson, K. S., Vester, B. M., Apanavicius, C. J., Kirby, N. A., & Schook, L. B. (2009). Implications of age and diet on canine cerebral cortex transcription. Neurobiology of Aging, 30, 1314—1326.
-
Benno, Y., Nakao, H., Uchida, K., & Mitsuoka, T. (1992). Impact of the advances in age on the gastrointestinal microflora of beagle dogs. Journal of Veterinary Medical Science, 54(4), 703—706.
-
Gorbach, S. L., Nahas, L., Lerner, P. I., & Weinstein, L. (1967). Effects of diet, age, and periodic sampling on numbers of fecal microorganisms in man. Gastroenterology, 53(6), 845—855.
-
Hopkins, M., Sharp, R., & Macfarlane, G. (2001). Age and disease related changes in intestinal bacterial populations assessed by cell culture, 16S rRNA abundance, and community cellular fatty acid profiles. Gut, 48(2), 198–205. doi:10.1136/gut.48.2.198
-
Calvani, R., Picca, A., Lo Monaco, M. R., Landi, F., Bernabei, R., & Marzetti, E. (2018). Of microbes and minds: A narrative review on the second brain aging. Frontiers in Medicine, 5, 53. doi: 10.3389/fmed.2018.00053
-
Rampelli, S., Candela, M., Turroni, S., Biagi, E., Collino, S., Franceschi, C., Brigidi, P. (2013). Functional metagenomic profiling of intestinal microbiome in extreme ageing. Aging (Albany NY), 5(12), 902–912. doi: 10.18632/aging.100623
-
Hoffman, J. M., Creevy, K. E., Franks, A., O’Neill, D. G., & Promislow, D. E. L. (2018). The companion dog as a model for human aging and mortality. Aging Cell, 17(3), e12737. doi:10.1111/acel.12737
-
Kaeberlein, M., Creevy, K. E., & Promislow, D. E. L. (2016). The Dog Aging Project: Translational Geroscience in Companion Animals. Mammalian Genome : Official Journal of the International Mammalian Genome Society, 27(7-8), 279–288. doi: 10.1007/s00335-016-9638-7
